12. Nucleophilic Participation During Electrophilic Addition to Alkenes
Freshman Organic Chemistry II (CHEM 125B)
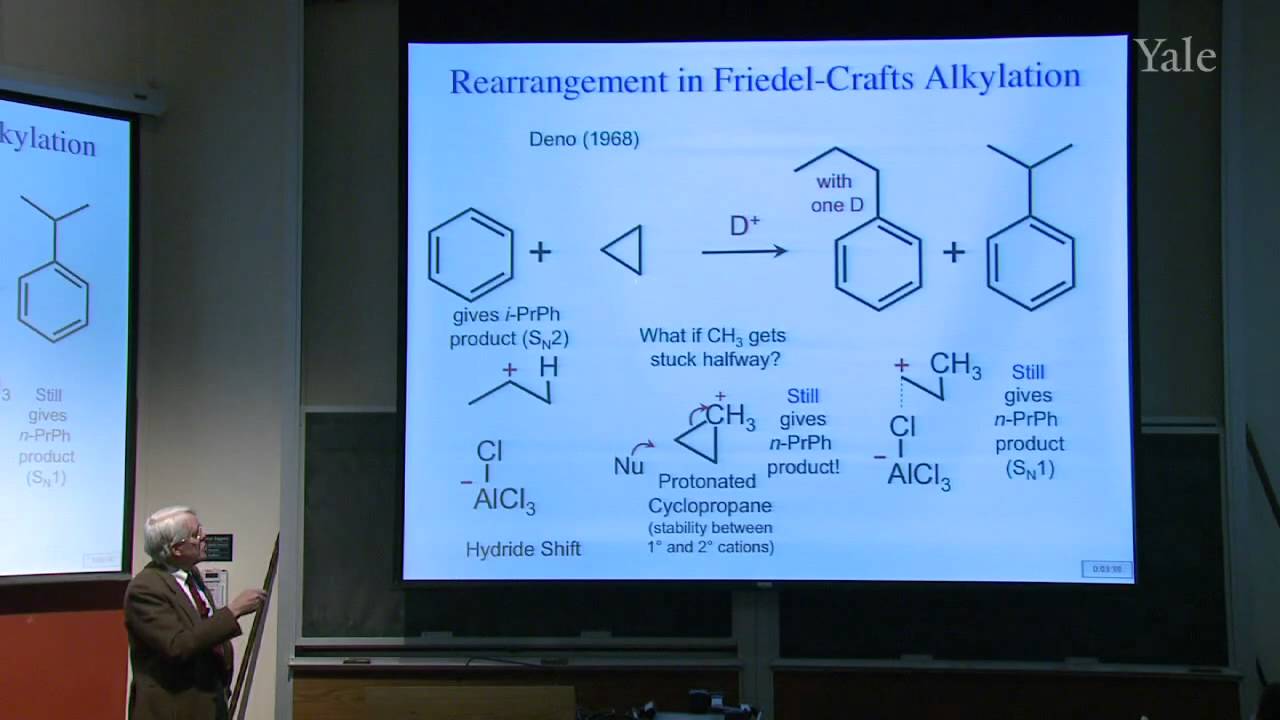
When electrophilic addition involves a localized carbocation intermediate, skeletal rearrangement sometimes occurs, but it can be avoided when both alkene carbons are involved in an unsymmetrical 3-center-2-electron bond, as in Markovnikov hydration via alkoxymercuration followed by reduction. Similarly a reagent that attacks both alkene carbons simultaneously by providing a nucleophilic component during electrophilic attack can avoid rearrangement, as in reactions that proceed via three-membered-ring halonium intermediates. Simultaneity in making two bonds during formation of cyclopropanes from carbenes can be demonstrated using stereochemistry. Anti-Markovnikov hydration can be achieved via hydroboration followed by oxidation with hydroperoxide. Rearrangement of the borane hydroperoxide intermediate with frontside C-O bond formation shows close orbital analogy to backside attack during SN2 substitution. Again syn-addition shows that nucleophilic attack occurs simultaneously with electrophilic attack on the alkene.
00:00 - Chapter 1. Forming Unrearranged Alcohols via Hydroxymercuration
07:36 - Chapter 2. Electrophilic Addition to Alkenes with Nucleophilic Participation: Halonium Ions
24:56 - Chapter 3. Electrophilic Addition to Alkenes with Nucleophilic Participation: Carbenes
36:55 - Chapter 4. Anti-Markovnikov Hydration via Hydroboration and Oxidation
Complete course materials are available at the Open Yale Courses website: http://oyc.yale.edu
This course was recorded in Spring 2011.































![35. Acyl Insertions and [gr]α-Reactivity](https://i.ytimg.com/vi/Xb4CwzP-Hdk/maxresdefault.jpg)
![36. [gr]α-Reactivity and Condensation Reactions](https://i.ytimg.com/vi/DQfUaXLk_sw/maxresdefault.jpg)