31. Periodate Cleavage, Retrosynthesis, and Green Chemistry
Freshman Organic Chemistry II (CHEM 125B)
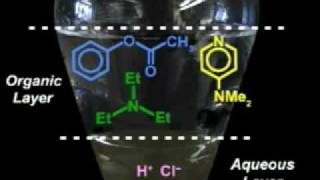
The ability of periodic acid (HIO4) to cleave the C-C bond of vicinal diols and [gr]α-hydroxycarbonyl compounds allowed structure determination of sugars and their ketals before spectroscopy was available. Reduction of carbonyl compounds by organometallic or hydride reagents provides a range of schemes for synthesizing various alcohols, where preference may be dictated by the desire to avoid competing processes. Wittig olefination allows conversion of C=O to C=C with good control over constitutional isomerism. Pharmaceutical manufacturers have taken great interest in developing new solvents and reagents that minimize hazards, waste, and environmental impact of traditional reactions.
00:00 - Chapter 1. HIO4 Vicinal Diol Cleavage and Traditional Carbohydrate Analysis
09:32 - Chapter 2. Designing Alcohol Syntheses
28:35 - Chapter 3. Addition, Reduction, and Enolization by Grignard Reagents
33:15 - Chapter 4. Wittig Olefination
37:45 - Chapter 5. What Green Chemistry Needs
Complete course materials are available at the Open Yale Courses website: http://oyc.yale.edu
This course was recorded in Spring 2011.